Miravirsen (INN; codenamed SPC3649) is an experimental drug for the treatment of hepatitis C, being developed by Santaris Pharma. As of 2017 it was in Phase II clinical trials.[1]
Miravirsen had been given by subcutaneous injection in early clinical trials as of 2017.[1] It is antisense to a human microRNA called miR-122. miR-122 ferries an argonaute protein to 5'-UTR region of viral RNA, where it binds, protecting the RNA from being destroyed by normally present nucleases; by binding to miR-122, miravirsen removes that protection and the virus RNA can be destroyed.[1] There is some evidence that the 5'-UTR region mutates under repeated exposure to miravirsen.[1]
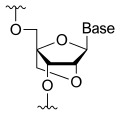
Miravirsen is a modified oligonucleotide consisting of a chain of 15 nucleotides, the base sequence of which is designed to selectively bind to miR-122.[1][2] Seven of the 15 sugar units are deoxyriboses, and the other eight are riboses with an additional bridge between the 2' oxygen and the 4' carbon atoms; this makes the molecule a locked nucleic acid. Furthermore, the phosphate units have been replaced by thiophosphates.[2]
The complete base sequence is
mC*-dC-A*-dT-dT-G*-mU*-dC-dA-mC*-dA-mC*-dT-mC*-mC*[d= 2'-deoxy,m= 5-methyl,*= 2'-O,4'-C-methylene, i.e. bridged or "locked" sugar]
with 3'→5' thiophosphate linkages.[2]









You must be logged in to post a comment.