Zirconyl chloride is the inorganic compound with the formula of [Zr4(OH)8(H2O)16]Cl8(H2O)12, more commonly written ZrOCl2·8H2O, and referred to as zirconyl chloride octahydrate. It is a white solid and is the most common water-soluble derivative of zirconium. A compound with the formula ZrOCl2 has not been characterized.[1]
Production and structure
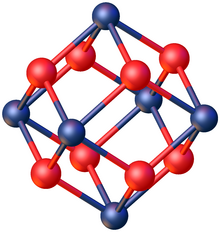
The salt is produced by hydrolysis of zirconium tetrachloride or treating zirconium oxide with hydrochloric acid.[2] It adopts a tetrameric structure, consisting of the cation [Zr4(OH)8]8+. features four pairs of hydroxide bridging ligands linking four Zr4+ centers. The chloride anions are not ligands, consistent with the high oxophilicity of Zr(IV).[1] The salt crystallizes as tetragonal crystals.[3]
See also
References
- ^ a b Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 978-0-08-037941-8.
- ^ Ralph Nielsen "Zirconium and Zirconium Compounds" in Ullmann's Encyclopedia of Industrial Chemistry, 2005, Wiley-VCH, Weinheim. doi:10.1002/14356007.a28_543
- ^ T. W. Mak "Refinement of the crystal structure of zirconyl chloride octahydrate" Canadian Journal of Chemistry, 46, 3491 (1968) doi:10.1139/v68-579









You must be logged in to post a comment.