User talk:Benjah-bmm27: Difference between revisions
dont abandon us |
Shootbamboo (talk | contribs) →odd looking chembox: new section |
||
| Line 1,060: | Line 1,060: | ||
==Well dont abandon us!== |
==Well dont abandon us!== |
||
Other editors must share your sense of exasperation over the persistent irresponsible/nutty behavior. You have to admit that most pages are much better than when you and I started. Fewer real embarrassments. Readers now must understand that most inorganic solids have polymeric structures. So for the annoying stupidity, including university-driven content, we just grind away at the worse parts when these editors cease at the end of their term. So dont lose hope. And be resigned to the fact that we're always going to have this kind of strangeness.--[[User:Smokefoot|Smokefoot]] ([[User talk:Smokefoot|talk]]) 18:02, 16 April 2011 (UTC) |
Other editors must share your sense of exasperation over the persistent irresponsible/nutty behavior. You have to admit that most pages are much better than when you and I started. Fewer real embarrassments. Readers now must understand that most inorganic solids have polymeric structures. So for the annoying stupidity, including university-driven content, we just grind away at the worse parts when these editors cease at the end of their term. So dont lose hope. And be resigned to the fact that we're always going to have this kind of strangeness.--[[User:Smokefoot|Smokefoot]] ([[User talk:Smokefoot|talk]]) 18:02, 16 April 2011 (UTC) |
||
== odd looking chembox == |
|||
At [http://en.wikipedia.org/wiki/2,5-Dimethoxy-4-iodoamphetamine DOI] the infobox looks odd under the Jmol images section. A quick look from me didn't spot anything. Might be a quick fix can you take a look? Thanks. [[User:Shootbamboo|Shootbamboo]] ([[User talk:Shootbamboo|talk]]) 00:12, 17 April 2011 (UTC) |
|||
Revision as of 00:12, 17 April 2011
Template:Archive box collapsible
I saw the structure. So its the zwitterionic tautomer that crystallizes? Makes sense. If so, I'll redo the ChemDraw, perhaps showing an equilibrium. --Smokefoot (talk) 01:28, 14 January 2009 (UTC)
- Apparently so, here's the ref: Acta Cryst. (1996). C52, 1681-1683. I agree with your suggestions, so I'm starting to upload unit cells, coordination polyhedra and extended arrays for polymeric materials. I'm working on YBCO right now - see Commons:Category:Yttrium barium copper oxide.
- All the best
C2O5-anion
This template must be substituted, see Template:Smile for instructions
... http://en.wikipedia.org/w/index.php?title=Talk:Triethyloxonium_tetrafluoroborate&diff=266749919&oldid=266749677 .. --Dirk Beetstra T C 14:50, 27 January 2009 (UTC)
Ben, Thanks for Replying
Ben, I'm still quite uncertain about navigating on Wikipedia. But I have learned a lot about editing.
No, I didn't think you were concerned about that changed name for the Image file for my Figure 3. What happened there was that I had found a minor error in the drawing and thus made a new one on my pc. But in uploading it into Commons, all the hyphens in the original drawing were replaced by underscores (actually a good thing since then one could bring in the revised drawing without bringing in the old one again. But it too me a brief while not to understand that I had to change all the hyphens in the Edit copy for the drawing to underscores. And that solved the problem.
My only comment about the minus sign was that I didn't want you to think that it my part of the Birch article where you found an error (at least I think so). The history of that article is that I had it in my User Space. I had written it when I noted that the Birch article Wikipedia had was very limited and had some errors which only an organic chemist would find. First I did try making some minor corrections in the old one (mainly missing references) but that met resistance. So I wrote an entirely new one. Finally it was moved to Article Space and then merged with the old one. It was a part of the old one in which you found a problem with minus signs; at least I think so.
Ben, it was nice to hear from you. You are in England a place I saw back in 1944-45 on a troop train going from Glascow to Southampton. Of course, later I got to see England in much nicer times.
With best wishes,
--Howard E. Zimmerman 22:49, 20 April 2010 (UTC) —Preceding unsigned comment added by Hezimmerman (talk • contribs)
Carboxyl Group
Ben, Thanks for the heads up on the Carboxyl group geometry. Your work on wikipedia is impressive. I uploaded the new image to the page Carboxyl.
Itineranttrader (talk) 00:43, 3 February 2009 (UTC)
Thanks for the note
Thanks for the encouragement as well as the nifty structures. Even if we dont educate the world, we seem to be learning bits ourselves.--Smokefoot (talk) 01:26, 6 March 2009 (UTC)
Plumbide
I noticed that you changed the formatting for Plumbide slightly, putting the links to 18-crown-6 and en to the sentence after the chemical structure rather than in the chemical structure itself. Why is that? Chlorine Trifluoride (talk) 14:34, 6 March 2009 (UTC)
- For clarity - we don't normally put links inside chemical formulae.
- The guideline is at Wikipedia:Manual of Style (chemistry)#Reactions.
What do you think of the info that I added from Greenwood and Earnshaw? Chlorine Trifluoride (talk) 14:12, 8 March 2009 (UTC)
- Very good. Would you like me to add an image from the crystal structure of one of these compounds?
- Yeah. I noticed that there are pictures in the book, but I don't know if we can use those. Chlorine Trifluoride (talk) 14:28, 8 March 2009 (UTC)
- Don't worry, I'll make some in 3D from the original data.
- Does the image you posted apply to all nonaplumbide anions or just the potassium cation one? Chlorine Trifluoride (talk) 14:08, 9 March 2009 (UTC)
- I don't know. Greenwood & Earnshaw was written before the nonaplumbide ion had been crystallographically investigated, so it does not discuss the structure (other than to say it's unknown, p. 394). Try a review article - I'll keep looking, too, and let you know.
I found "Aspects of anionic framework formation: Clustering of p-block elements". doi:10.1016/S0010-8545(98)00168-4. Chlorine Trifluoride (talk) 15:20, 10 March 2009 (UTC)
Additionally, can you also do the structure for the pentaplumbide anion? Chlorine Trifluoride (talk) 16:19, 12 March 2009 (UTC)
- No problem. The image is File:Pentaplumbide-anion-from-xtal-3D-balls.png.
- Cheers
Hexaphenylcyclotrisiloxane and octaphenylcyclotetrasiloxane
Hi Ben. I noticed that you have recently uploaded nice structures of cyclic siloxanes substituted with phenyls. They are not used in an article as far as I see. Do you know, if hexaphenylcyclotrisiloxane and octaphenylcyclotetrasiloxane have industrial or commercial applications (i.e. they are produced/used in significant amounts)? --Leyo 17:41, 17 March 2009 (UTC)
- I don't know, really. I just thought they were interesting examples of conformation. I'll have a look in textbooks.
Isobutane
I have not removed galleries from articles, but a gallery from an article. I acknowledge that there really is no problem. I will explain more in depth on Talk:Butane. Happy Wiki'ing! Bobber0001 (talk) 10:32, 18 March 2009 (UTC)
- Sorry about the plural, I thought it was more than one article. No problem though, happy editing :)
Hi, next time please be careful when reverting edits, as you did with User:Ratpole's edit to Gold. One of his/her changes included a valid spelling correction (abandonded -> abandoned), which you also reverted. I've already fixed the problem, but just keep an eye out for this in the future. Thanks!—Tetracube (talk) 22:58, 8 April 2009 (UTC)
- OK, sorry about that. It is difficult, though. Probably easier to just revert the lot then fix abandonded. It all works out in the end.
- Thanks
Molecule structures
Hi, Ben. Thanks for the encouraging message. I actually got started making the 3D molecules using your tutorial, so in a way you are sort of my mentor. I would love some pointers on where to find the correct geometries for different molecules so that I do not make the mistake I made again. Thanks! Woodenchemist (talk) 16:11, 10 April 2009 (UTC)
- You're doing a great job. I frequently run into trouble trying to make images that turn out to be misleading - but I learn a lot of chemistry along the way!
- If you have access to textbooks, journals and search engines like SciFinder, that'll make things much quicker. If not, there's plenty of free information on the web. The basic idea is simple, and is the same as writing text for Wikipedia: you need to provide a reliable source with each image. The places I often get my structural information from are:
- textbooks that show bond lengths and angles, such as Greenwood & Earnshaw
- journal articles - structures often determined by X-ray diffraction, microwave spectroscopy or electron diffraction
- there are two good websites that publish crystal structures electronically: the AMCSD is free but has mostly minerals, whereas the IUCR's journals have all sorts of compounds, but only some of their content is open-access, notably Acta Crystallographica Section E: Structure Reports Online
- the CRC handbook has a useful table of the gas-phase structures of small molecules
- It's normally not good to build a 3D structure from the 2D formula given in a Wikipedia article, because there can be important aspects of the molecule's structure (such as its conformation) that might be calculated wrongly by DS Visualizer.
- If you'd like me to find and send you any structural information that you're finding hard to track down, or if you'd like any more tips, just drop by!
- Cheers
Re: Thallium iodide
Dear Ben, you were right to revert my edit of TlCl. Indeed, it is CsI-type at room temperature and converts to TlI structure at low-T only. I have put references on TlI structure as (A-V Mudring "Thallium Halides - New Aspects of the Stereochemical Activity of Electron Lone Pairs of Heavier Main-Group Elements" Eur. J. Inorg. Chem. 6 (2007) 882)(R. P. Lowndes and C. H. Perry "Molecular structure and anharmonicity in thallium iodide" J. Chem. Phys. 58, 271 (1973)) I can email you these pdfs if you wish.
Regarding ZnO. IMO, the images are redundant because  is not the unit cell, but its expanded graphical depiction, i.e. just a cropped version of
is not the unit cell, but its expanded graphical depiction, i.e. just a cropped version of  . The wurtzite unit cell contains only 4 atoms (see, e.g. http://portellen.phycmt.dur.ac.uk/sjc/thesis_mcg/node47.html). Also, for most readers, unit cell does not say much, i.e. one structural picture is sufficient. If you agree, you may wish to revert your edit, but this of no importance. Best regards.NIMSoffice (talk) 00:19, 23 April 2009 (UTC)
. The wurtzite unit cell contains only 4 atoms (see, e.g. http://portellen.phycmt.dur.ac.uk/sjc/thesis_mcg/node47.html). Also, for most readers, unit cell does not say much, i.e. one structural picture is sufficient. If you agree, you may wish to revert your edit, but this of no importance. Best regards.NIMSoffice (talk) 00:19, 23 April 2009 (UTC)
- I agree, its not a major issue. These diagrams are fairly difficult to interpret unless you know a few unit cells, and you're likely to know wurtzite and sphalerite if you know any!
I looked up the definition of unit cell, which I have forgotten actually. Loosely speaking, most of those ball-and-stick models are unit cells (thus no need to put several, I usually choose one which looks clearer). More restrictive (thought not unique too) is "primitive unit cell", i.e. the smallest number of atoms to build the lattice (e.g. 4 for ZnO). Those are however not so illustrative. NIMSoffice (talk) 03:34, 23 April 2009 (UTC)
- Yes, you're absolutely right. If I had software that could do it, I'd make unit cells with sliced-through atoms at the corners, e.g. http://www.wou.edu/las/physci/ch412/unitcel1.jpg, http://www.cbu.edu/%7Emcondren/NaCl-unit-cell.jpg, http://departments.kings.edu/chemlab/vrml/reox.html.
Boat conformation
Hi Ben. Could you draw a boat conformation version of File:Cyclohexane-chair-2D-stereo-skeletal.png? The version in Commons:Category:Cyclohexane conformation missed the perspective. I tried to draw one, but as it didn't look nice, it didn't upload it. --Leyo 12:42, 6 May 2009 (UTC)
- No problem, here it is: File:Cyclohexane-boat-2D-stereo-skeletal.png.
- Cheers
- Ben (talk) 22:52, 6 May 2009 (UTC)
- Great, thanks. --Leyo 13:01, 7 May 2009 (UTC)
Fluoxetine structure
Hey Ben, you may want to weigh in here: Talk:Fluoxetine#Question on the Chemical Structure of Fluoxetine. Best, Fvasconcellos (t·c) 13:08, 6 May 2009 (UTC)
Boron nitride
Dear Ben, I am working on boron nitride hoping to bring it to GA level. The article desperately needs pictures. Would you be willing to draw them ? For example, something like boron-nitride nanotube (same as carbon nanotube, but with alternating boron and nitrogen atoms) and intercalated hexagonal BN. Intercalation of BN is difficult, and thus I am trying to be careful in selecting credible sources. I trust Solozhenko[1] and he has reported BNXNH3 with x=3,4,5 where NH3 molecules are between the BN sheets (no picture given). Another intercalation is alkali metals (with a picture).[2] Any other ideas on pictures and contents are welcome. Cheers.NIMSoffice (talk) 06:51, 13 May 2009 (UTC)
- ^ V. L. Solozhenko (2002). "In situ studies of boron nitride crystallization from BN solutions in supercritical N–H fluid at high pressures and temperatures". Phys. Chem. Chem. Phys.: 5386. doi:10.1039/b206005a.
- ^ G. L. Doll; et al. (1989). "Intercalation of hexagonal boron nitride with potassium". J. Appl. Phys. 66: 2554. doi:10.1063/1.344219.
{{cite journal}}: Explicit use of et al. in:|author=(help)
- Hi NIMS, I'll be delighted to make pictures for boron nitride. I've got exams next week and the week after, but I may be able to draw a few during that time.
- I'll check the crystallographic databases for relevant structures, and follow your links.
- Cheers
I had a look on the crystallographic databases I have access to, but could not find any BN intercalation compounds. I also read the papers you link to above – the 1989 paper didn't seem to identify the interlayer separation, and I am not advanced enough to really understand their reciprocal lattice diagram. `The BN-NH3 paper didn't seem to contain any coordinates.
Ben (talk) 10:41, 7 June 2009 (UTC)
- Ben, sorry for deleting this section, refs were wrong, but I fixed them. I have drawn ref. 2 in boron nitride (found cell parameters), but not ref. 1; it might not be needed anymore. I also deleted your drawing (HgS I guess, you restored it, and its fine), again, without harm intended - just thought of redundancy. You might have noticed that I'm much too quick on action :) I've been drawing much (minerals, etc.) these days. Cheers. Materialscientist (talk) 10:46, 7 June 2009 (UTC)
Not a problem, I just like to keep a record of what's been said. It was HgO, I think. I looked into the polymorphs of HgO and there are many - see User:Benjah-bmm27/Task list. I'll add pictures shortly. Don't worry about redundancy, there's plenty of room in Wikipedia, the most important thing is to convey the information clearly.
I have a few tips for your drawings, such as File:OsO4structure.jpg:
- upload them to the Wikimedia Commons - then other language Wikipedias can use them, too
- make your images as PNGs, not JPEGs - JPEG suffers from worse compression artifacts
- write the source of the structural information (I see you are starting to do this) and the software used to create the image in the image description template
- use a standard colour coding scheme for your atoms, such as this one I use
- a white or preferably transparent background is neater
- as well as the unit cell, e.g. File:Domeykite_structure.jpg, it's helpful to give an orthographic view (no perspective) along one of the main symmetry axes of more than one unit cell, to show the periodicity and packing in the crystal, such as this (also see below):
-
GaF3 -
As2O5
Ben (talk) 11:08, 7 June 2009 (UTC)
- Ben, thanks for advice. I did not think about saving in PNG; though I could do that. I know about WP commons, but after changing username, my WP and Wikimedia accounts got somewhat unconnected. I've fixed that recently and can move images to commons as I've done before. One reason I prefer WP is more control over captions, etc. - on wikimedia I often get to know that they want to delete my image only at the last moment. Another good advice is to plot several projections. I didn't do that because for many articles more than one structural image is too many, though in some cases several projections could be necessary. I can recreate them easily. That comes back to HgO - many compounds have too many crystal forms; drawing them all is easy, but would overload the article. Black background is a glitch of one of the programs I use (still don't know how to change it easily, but the program is very, very handy in other respects). Atomic colors are stable in those black pictures. I usually mention the color in figure description. If you notice I missed that, please fix (I've just done that for one of yours). File:OsO4structure.jpg is a tricky example - very low symmetry. You're right about File:Domeykite_structure.jpg, but the structure is hard to grasp anyway. The only reason for plotting that picture is to show that the structure could be complex even for cubic cell (I think I mentioned it in the picture description; just reconfirmed - that structure is same bloody mess in any possible projection).
- Coming back to your drawings, IMHO small balls + sticks are much better than ball-packing structures - there is no need to know "electron clouds" and the like. Have a look at Graphite intercalation compound and see for yourself (well, I should have added lines for the Ca sublattice too). Materialscientist (talk) 11:38, 7 June 2009 (UTC)
You could take your images with a black background and remove it using Photoshop or GIMP. You're probably right about OsO4, although it is possible to show a bit of packing. Not necessary though, molecular structures are mostly about the molecule! I respectfully disagree that space-filling models are less desirable than ball-and-stick models. Graphite intercalation compound is a prime example! You can see the way the potassium atoms prop open the graphite layers. Both styles are complementary - although in some cases, one may be much clearer.
Thanks for adding the reference to File:Rutile-unit-cell-3D-balls.png. I've updated the image description. I think it's important to give the source that was actually used for the image, rather than any convenient reference that seems to say the same thing. Sometimes there are differences or mistakes in sources that we don't notice until later.
You make a good point about noting atom colours explicitly. I will start to do that in my image descriptions. It's not always practical, though, to add a caption. Chemboxes are a notable example. And often, it is easy to deduce which atom is which colour, e.g. phosphorus pentafluoride!
Ben (talk) 12:20, 7 June 2009 (UTC)
Tetramethylammonium chloride
Dear Ben, I saw your new structure - replacing my one :-( c'est la vie. My one question is that which of us is correct? I set IsisDraw up according to Wikipedia:Manual of Style (chemistry)/Structure drawing, and I got the result that I posted. Personally I prefer your structure - I always do Hydrogen "on Hetero and Terminal" at work - but the WP page says just "on Hetero" Ronhjones (Talk) 23:34, 19 May 2009 (UTC)
- Hi Ron, your image is factually correct, but it has several drawbacks:
- low resolution (small)
- unrealistic 90° CNC bond angles
- hidden (implied) hydrogens on methyl groups are confusing, especially in a structure that is otherwise not skeletal, as in this case
- Hydrogens are not always hidden, see Wikipedia:Manual of Style (chemistry): "Hydrogens should be implied (hidden), except for the benefit of the target audience" - in fact, Wikipedia chemists prefer explicit methyl groups - this should be explained in the MoS, I will raise the issue
- Thanks for asking, your questions will help improve the MoS and I'm sure you'll soon be up to speed with WP:CHEM's preferred styles.
- Thanks for the reply. As I said, I do prefer to see a methyl as a CH3. As for the drawing, well you have better software than me. IsisDraw does not excel in such nice diagrams. As for the size, well I followed the MoS, loaded up the JACS template, adjusted settings as described, made structure, and did a copy and paste into MSpaint, and then saved it as PNG (that really determined the size of 90px). There's no much else one can do, certainly the export into tiff (within Isis) fails - you get a tiff, which is OK on the PC, and when uploaded to WP, it not visible!
- As an aside, I saw on a Symyx e-mail at work (we use Beilstein Crossfire via Symyx's Discoverygate) that Symyx Draw 3.2 is now free (they also give away IsisDraw 2.5), just dowloaded that - it's a 63MB zip file (one has to register at www.symyx.com, but there is no charge for a private user). It's has a lot in common with Isis, but it does allow one to save the image directly as a PNG. I may still use Isis and finish off in Symyx though as I can draw very quickly in Isis. Ronhjones (Talk) 20:57, 20 May 2009 (UTC)
I'm pretty sure it is possible to get IsisDraw to export a bigger image. I haven't used it for a few years, but I believe other Wikichemists do. Post a message at Wikipedia talk:WikiProject Chemistry - people there are normally very helpful. You'll be a welcome addition to the gang. Let me know if there's anything else you need pointers on.
Cheers
Ben (talk) 06:21, 21 May 2009 (UTC)
Once you get a TIFF, use software like IrfanView to convert it into a PNG, at the maximum resolution possible. Typically 720 dpi or similar. --Rifleman 82 (talk) 16:23, 11 June 2009 (UTC)
- Actually, I might have found another way - I draw the molecule in Isis (or Symyx) Draw, then export as a MolFile. I then use "mol2Svg_1_0" (see ref#1 on Chemical file format) to convert directly to SVG - after all a MolFile is a form of a vector graphic, seems mad to take that, save as PNG (raster), and try to get back to vector. I just did this with the image on O-Acetylpsilocin - the downside? = it won't do terminal methyls, and the hetero hydrogens don't always come where you want. I've not yet tried to see if I can "tweak" the final image with Inkscape. Ronhjones (Talk) 00:24, 12 June 2009 (UTC)
Thallium(I) bromide
Ben, why reverting ? Why two same figures ? Pride for own creation ? There are so many wrongs there. To start with, red and brown colors are no good couple. Atoms do not pack simply because their ionic/covalent "radii" is a primitive abstraction developed around 1920 when no better methods were available. This abstraction proportionally splits a distance between atoms in a solid, names it "radius" and assumes the electron shell is a sphere having that radius. By definition, it gives dense packing, but. We know that bonding electron distribution is very far from spherical. Another point is that all structural models we use are crude approximations, but dense packing is graphically poor to show relative atomic positions, as compared to ball-stick models. Materialscientist (talk) 23:20, 11 June 2009 (UTC)
- No, not pride! All the things you say are true, but these depictions are commonplace and useful pedgogically.
- I don't really mind what the conclusion is, but discuss at WP:CHEM first - you're making a lot of unilateral edits quickly.
Hello,
I just wanted to mention that the File:Fluorite-unit-cell-3D.png is not to scale. I've added a warning in the image description to tell that, because I have made the error to exchange F and Ca atoms based on this image (I should have check more carefully)... --Mathieu Perrin (talk) 15:17, 11 June 2009 (UTC)
- I think you might be mistaken in this case - I used ionic radii in the image, since fluorite is predominantly an ionic compound: r(Ca2+) = 1.00 Å, r(F−) = 1.33 Å.
- You're right !! I'm using only covalent crystals usually, so I forgot to use ionic radius... I still have a distance problem, however. Using your values, I find
- for contacts between spheres as shown in the figure, and
- by assuming contact between F ions.
- There is a certain deviation to the exp. 5.46 Å value... ?? Maybe not significant enough. I've changed my warning into "information" in the figure. Which software are you using for modeling? --Mathieu Perrin (talk) 14:42, 15 June 2009 (UTC)
Ionic radii are averages over many crystal structures - they're not exact because ions are not perfectly hard spheres, there is some degree of covalency in even the most ionic of compounds. Differing degrees of covalency in different compounds would give rise to slightly different ionic radii if they were calculated separately for each compound.
This isn't normally a problem in images, as the picture would look almost identical, whether r(F−) = 1.33 Å or 1.35 Å etc.
Ben (talk) 14:49, 15 June 2009 (UTC)
Ben, could you please check File:Iron(II)-sulfide-unit-cell-3D-balls.png. My database tells this is exactly the structure of Ln2O3 (Ln=rare earths - pity I did not see that yesterday when I drew this Ln2O3 structure myself), and gives a quite different structure for FeS (mackinawite). Suspicious is also the ratio of atoms in the unit cell. Cheers. Materialscientist (talk) 00:40, 5 July 2009 (UTC)
- There are 2 Fe and 2 S in that unit cell. It is the NiAs structure, see http://www.benjamin-mills.com/chemistry/structures/NiAs/. I think there are several polymorphs of FeS. Check http://rruff.geo.arizona.edu/AMS/amcsd.php. Ben (talk) 13:47, 5 July 2009 (UTC)
I just saw this (I though I was watching this page, but I wasn't). I was wrong and your structure is correct. The similarity between File:Iron(II)-sulfide-unit-cell-3D-balls.png and File:La2O3structure.jpg, although the stoichiometries are different, is accidental - neither structure is a primitive cell. Indeed, FeS has many polymorphs, the information on that is obscure (because not only structure but also Fe/S ratio may change slightly, say 10-20% this is not very common and is misleading), but I might sort that information out later. Materialscientist (talk) 04:23, 8 July 2009 (UTC)
- Yeah, nonstoichiometric compounds are a nightmare to draw.
- Thanks for your continued hard work on simple solids, keep it up!
Drawing is easy - I have good access to structure files (but the program which can treat them is crippled - thats why black backgrounds). Most time is gone to understand the polymorphs. I also still can't figure out my PNG problem (my PNGs/SVGs display on WP only in full size, but not upon thumb reduction) - thats why still jpegs. You were right about Zirconium(IV) chloride, I forgot about similar layered structures (in fact, many different binaries have structures which are almost same graphically, but not crystallographically). Materialscientist (talk) 11:32, 8 July 2009 (UTC)
Ben, I have added the symmetry and phase transition for that article. Could you please fix the captions (if you remember) in terms of which structure (orthorhombic or tetragonal (it might be called "square" or "cubic" in some papers)) is shown there. Thanks. Materialscientist (talk) 00:36, 15 July 2009 (UTC)
- Done. The structure was taken from (I think) J. Solid State Chem. (1975) 13, 252-257.
Magnus Green Salt
Hi Ben
Could you do a picture for Magnus' green salt, because the one there sucks :). Ta, Chris (talk) 08:32, 17 July 2009 (UTC)
- No problem :)
Thanks. Chris (talk) 12:19, 17 July 2009 (UTC)
Molecular formula layout in chembox
Hi Benjah-bmm27,
Regarding [1], I don't dispute your edit, but I was wondering whether we could include a note at Wikipedia:Chemical_infobox or Template:Chembox_Properties stating the consensus? Even a link to a relevant guideline would be useful. I've looked both before and since making my edit, but I can't find any discussion about how these formulae should be presented, and I'm sure I'm not the first person to wonder. Adrian J. Hunter(talk•contribs) 13:31, 27 July 2009 (UTC)
- Absolutely, good idea. You can ask at WP:CHEM, or I can do it, but I will probably take a little time as something's up at home.
- Thanks for your response. I've started a thread at Wikipedia_talk:WikiProject_Chemicals#Nomenclature_for_molecular_formulas_in_Chembox. Adrian J. Hunter(talk•contribs) 14:03, 6 August 2009 (UTC)
3D Models
Just a question about models. How do you get the background of the image to be transparent? Thanks. Ginogrz (talk) 04:27, 31 July 2009 (UTC)
- I use Photoshop to remove the background - it's often helpful to pick a colour other than white so that you don't remove bits of hydrogen atoms in the process.
- Alright, I followed your instructions as well as I could and here's my first attempt. [2] The color of the carbon atoms doesn't seem the same as your models and the red of the oxygens definitely seems less vivid. I'm not sure why this is. Any observations and/or criticisms would be appreciated. Ginogrz (talk) 06:48, 1 August 2009 (UTC)
- I changed the colour palette to make carbon a much darker grey than it is by default — about 56% R, 56% G, 56% B, or #383838 or #484848.
- I didn't change the red of oxygen, though. If you tweak the lighting settings a bit, maybe move the darker light in the bottom right hand corner down a bit, things should brighten up.
- One thing I know can cause a little inconsistency is that I used version 1.5 but later versions have a slightly different look to them. It's not a problem, though, your image already looks smart.
- I also tend to make the hydrogen atoms a little smaller than all other atoms, by going Edit > Select > Atom... > H then ctrl+D and setting the atom radius to 0.3 (I think). This is purely a matter of choice but can often make a structure look less cluttered when many hydrogens are present.
- I would simply carry on as you are. The most important thing is to find articles that would benefit from illustrations, and use experimentally-determined structures where possible. This is because conformation is often chemically important and our own sense of structural intuition can often deceive us. Take disulfiram for example. I thought it would probably have a planar C2NC(=S)SSC(=S)NC2 core, but Acta Cryst. (1989). B45, 65-69 and Acta Cryst. (1986). C42, 1420-1423 indicate the CSSC dihedral angle is about 90°, i.e. not at all planar.
- I revised that model I linked, I think it looks pretty good. I like your settings much better. Thanks for the advice. I will take into account the importance of the experimental data as well. Thanks a lot. Ginogrz (talk) 05:48, 2 August 2009 (UTC)
Ca gluconate etc
Ben, thanks for the note about my claim that calcium gluconate is mis-drawn. It was a guess based on the nakedness of the Ca2+ center and the bis(bidentate) carboxylate (I have never seen that motif in acetates), the absence of aquo ligands, and the fact that the thing looks molecular (also rare for carboxylates). Possibly the picture is of a subunit in a polymer that forms upon dehydration. I would expect something far more complicated with octahedral Ca2+. But I havent gone digging yet in the literature.--Smokefoot (talk) 22:13, 18 August 2009 (UTC)
- Thanks for bringing that one to my attention - I've put quite a few dodgy structures out there in Wikiland. I'm sure you're right about anhydrous calcium gluconate. In medicine (treatment of HF burns etc), it is apparently the monohydrate that is used: http://www.globalcalcium.com/01Calciumgluconate_02.htm.
- Ben (talk) 22:54, 18 August 2009 (UTC)
- Well you're doing so much good work so don't worry about my cranky comments. And for species like this, who knows whether the Xray is very relevant to the structure in soln? --Smokefoot (talk) 00:38, 19 August 2009 (UTC)
Talkback

You can remove this notice at any time by removing the {{Talkback}} or {{Tb}} template.
Ronhjones (Talk) 19:26, 19 August 2009 (UTC)
Hi Ben. Do you remember where you got the 101.7 pm from? The value cited from this book is 101.4 pm (see article in de-WP). --Leyo 06:38, 28 August 2009 (UTC)
- Greenwood & Earnshaw, I think - I don't have the book to hand, though, so I can't confirm that figure yet. The book you cite is from 1955, when spectroscopy was a little less precise than in 1997, when G&E 2nd ed. was written.
- However, the CRC handbook gives the dimensions of ammonia, determined by IR spectroscopy, as N-H (re) = 101.2 pm and ∠HNH (θe) = 106.7°.
- Ben (talk) 08:58, 28 August 2009 (UTC)
- Thanks for your reply.
The first edition of this book might be from 1955.The first was printed much earlier. The one cited in from 2007. :-) It is pretty much the standard for inorganic chemistry. As it is in German, you might not know it. --Leyo 15:45, 28 August 2009 (UTC)
- Thanks for your reply.
Oh, sorry - confusing wording "No preview available - 2007 - 2149 pages. This book has a more recent edition (1955)." - makes no sense!
I know Holleman & Wiberg, it's the graduate-level inorganic text here, too.
Shall we have a look in the literature?
Ben (talk) 15:49, 28 August 2009 (UTC)
- I found the statement in an earlier version of the same book on Google Books now. No original source is indicated there. --Leyo 15:59, 28 August 2009 (UTC)
HI..........
Hi....... I think we really need to do some work on the articles related to electropositivity, screening effect, etc... It seems to be very inadequate, as was exposed by the question on the Science Reference Desk.... Rkr1991 (Wanna chat?) 04:31, 1 October 2009 (UTC)
- Good idea - I'll have a think about it.
Sodium pertechnetate needs a new image
Hi, I noticed that you have done a lot of chemistry-related images. Could you help make a new image for Sodium pertechnetate? The current image is a low-resolution GIF; I think we can do better. :-) Thanks!—Tetracube (talk) 19:36, 25 November 2009 (UTC)
- Done: File:Sodium-pertechnetate-2D.png. Sorry for the epic delay!
And now... yes, you've guessed it
Chemical structure drawings
Hi, I followed your link from the reference desk to the structure of the ruthenium carbonyl hydride. I have a couple of questions,
- What do you use to draw chemical structures? (I assume you drew that one)
- Would it be possible to me to use that picture in my lab report if I properly reference it?
- If yes for the question above, would it be any trouble to have a picture of the structure of the Ru(CO)H2(PPh3)3 ruthenium carbonyl dihydride?
This is for a first year lab report and it is messy to hand draw in structures onto a typed report. Thanks for any help 188.221.55.165 (talk) 12:06, 12 January 2010 (UTC)
- Does User:Benjah-bmm27/MakingMolecules help to answer your first question? --Leyo 12:43, 12 January 2010 (UTC)
Hello,
Can you give a link to the picture in question, just so I can be sure it's one I made?
If it is, you can go right ahead and use it - all my images are in the public domain, so you don't actually need permission, but it's nice of you to ask nonetheless.
I'm not able to make an image of Ru(CO)H2(PPh3)3 in exactly the same style as my other images on Wikipedia, because I have changed software, but I could get a crystal structure and make it into an image in Jmol if you like.
Alternatively, you could just draw the molecules in ChemDraw or similar (in 2D) as this is the more usual representation used in most lab reports and academic papers - scientists usually only put 3D structures in when they have calculated or determined the structure themselves and the precise geometry of the molecule is the main topic of the report.
Ben (talk) 14:30, 12 January 2010 (UTC)
- Thanks for pointing to my tutorial, Leyo :)
I've made it into a 3D Jmol model for you, see if that's any use: http://www.benjamin-mills.com/chemistry/structures/Ru(CO)H2(PPh3)3/.
Ben (talk) 15:10, 12 January 2010 (UTC)
Cl2O7
- Electrons required for octets: 2*8+7*8=72
- Valence electrons available: 2*7+7*6=56: total pairs: 56/2=28
- Difference 72-56= 16: shared pairs (bonds): 16/2=8
- Difference 28-8= 20 lone pairs
Valence electrons available: 2*7+7*6=56: total pairs: 56/2=28
Difference 72-56= 16: shared pairs (bonds): 16/2=8
Difference 28-8= 20 lone pairs
- Thus, I think.
Greetings Jcwf (talk) 03:59, 19 January 2010 (UTC)
What software do you use for 2D diagrams?

Hello Benjamin, I must thank you for all the 2D and 3D molecular drawings that you provided for my Wikipedia articles. One could argue that your images are the best part of all chemistry articles.
I see that you have posted intructions about how to generate 3D images; thanks for that too. But what software do you use for the 2D "roadkill" diagrams, like that of cyclohexanehexone? I tried a couple of free downloads for Unix but could not get usable results from them. All the best, --Jorge Stolfi (talk) 18:39, 26 January 2010 (UTC)
- Hi Jorge,
- I like pictures so I make more of them! I use ChemDraw and then Photoshop to make my images, although many other free molecule editors and image processing software should be able to make identical images.
- If you need any pointers to get hold of suitable software, just let me know and I'll be happy to help
- p.s. nice work on the carbon oxides, you've added lots of interesting new content.
- Thanks about carbon oxides. (That subject was a 25-year-old unsnswered question of mine. Finally Wikipedia gave me the motivation to research it, and the answer turned out much more interesting and complicated that I could have ever imagined.)
- Indeed the free programs I tried do draw 2D diagram; but the details (font, placement, thickness, spacing, etc.) are nowhere as nice. But I will keep searching. All the best, --Jorge Stolfi (talk) 15:26, 27 January 2010 (UTC)
I think the details you need are here: Wikipedia:Manual of Style (chemistry)/Structure drawing.
Ben (talk) 17:43, 27 January 2010 (UTC)
Temporary blindness
Oops. I saw something bent, recalled vaguely you comments on non-bent Hg, knew some of these drawing come from organic chemists at one time, and assumed the worst... Thanks for not snapping at me. --Smokefoot (talk) 19:53, 3 April 2010 (UTC)
Speedy deletion nomination of Organyl group

A tag has been placed on Organyl group requesting that it be speedily deleted from Wikipedia. This has been done under section A3 of the criteria for speedy deletion, because it is an article with no content whatsoever, or whose contents consist only of external links, a "See also" section, book references, category tags, template tags, interwiki links, a rephrasing of the title, or an attempt to contact the subject of the article. Please see Wikipedia:Stub for our minimum information standards for short articles. Also please note that articles must be on notable subjects and should provide references to reliable sources that verify their content. You may wish to consider using a Wizard to help you create articles - see the Article Wizard.
If you think that this notice was placed here in error, you may contest the deletion by adding {{hangon}} to the top of the page that has been nominated for deletion (just below the existing speedy deletion or "db" tag), coupled with adding a note on the talk page explaining your position, but be aware that once tagged for speedy deletion, if the page meets the criterion, it may be deleted without delay. Please do not remove the speedy deletion tag yourself, but don't hesitate to add information to the page that would render it more in conformance with Wikipedia's policies and guidelines. Lastly, please note that if the page does get deleted, you can contact one of these admins to request that they userfy the page or have a copy emailed to you. &dorno rocks. (talk) 18:57, 13 April 2010 (UTC)
Nice graphics for chemical compound
Nice description and graphics on User:Benjah-bmm27/Gallery, User:Benjah-bmm27/MakingMolecules. Have you thought of saving your description of making 3D images on your website? In the end, wikipedia is a public website using CC-by-sa 3.0 license so even I can make use of your work. Ktsquare (talk) 17:48, 14 April 2010 (UTC)
- What's wrong with having User:Benjah-bmm27/MakingMolecules on Wikipedia?
- My comment is meant to be a suggestion so that letting you be aware of copyrights. I was attracted to your work and just giving you a heads-up. Ktsquare (talk) 21:53, 14 April 2010 (UTC)
- Oh OK, thank you. I was just checking what you mean, I didn't mean to sound snappy!
- I've already written the page and it's been on Wikipedia for years, so I don't think I would be able to claim back the copyright even if I wanted to. But since the text merely describes the use of a piece of free software to make images for Wikipedia, I'm ok with it being widely available.
- Thanks very much for your concern, though.
- Have you thought of joining Wikipedia:WikiProject Chemistry?
- Best wishes,
Barnstar
| The Template Barnstar | ||
| I award you this barnstar to show wikipreciation for the templates that you have created, keep up the good work! Jdrewitt (talk) 07:30, 16 April 2010 (UTC) |
- Thanks very much! I just made them to save a bit of time and provide consistency and metadata.
- Cheers!
Your Comments about a Minus Sign in the Birch Reduction
Benjah-bmm27 (Benjamin?)
I noted the comments about a minus sign in the Birch Reduction.
That seems to be in a part which I didn't write. I could be wrong but I think V8rik wrote that part.
After I wrote my Birch Reduction article it had parts of the Old Birch Reduction article merged with it.
There was a commentary about Figure 3 which I had difficulty bringing in to replace an earlier version which had one error. The problem turned out to be that the repaired drawing had a designation with underscores while the original version had hyphens and the title to the file had to be changed. But that should be unrelated to your comments.
Best wishes,
--Howard E. Zimmerman 18:23, 20 April 2010 (UTC)
- Dear Professor Zimmerman,
- Call me Ben.
- It's great to see a scientist of your stature helping out here on Wikipedia. You might like to join the Chemistry Wikiproject, where all the PhD chemists chew the fat. There are many highly qualified, talented, and friendly people there. The most interesting discussions take place at Wikipedia talk:WikiProject Chemistry and Wikipedia talk:WikiProject Chemicals
- Regarding my edit to the Birch reduction article, I wasn't implying any blame for the use of hyphen-minuses instead of minus signs, my edit summary was merely a description of the change I made.
- Thanks for all your hard work on the article, it's much better now.
- Cheers,
Transparency in D.S.
Hi Ben. I tried out Accelrys Discovery Studio 2.5 in order to replace incorrect molecule models on Commons. However, I was not able to save the image with a transparent background. On your how-to page you don't mention transparency at all. Could it be that Discovery Studio Visualizer 2.0 supports transparency, but Discovery Studio 2.5 doesn't? --Leyo 21:33, 29 April 2010 (UTC)
- I just use Photoshop to remove the opaque background. Most image editors should do this easily, a commonly used free one is GIMP.
- If you need any more help, just let me know :)
- All the best,
Chemicalinterest, Bismuth triiodide
I added that uncited example in bismuth triiodide because I thought that all metal oxides react with acids to form the corresponding metal salts. --Chemicalinterest (talk) 14:33, 11 May 2010 (UTC)
Sodium telluride
Could you make a visual diagram for sodium telluride? It would have the same shape as sodium sulfide, with the tellurium ions being a little bigger. --Chemicalinterest (talk) 21:00, 11 May 2010 (UTC)
- Never mind. I didn't know that a category existed that had all compounds missing a structure diagram.--Chemicalinterest (talk) 21:09, 18 May 2010 (UTC)
Talkback

You can remove this notice at any time by removing the {{Talkback}} or {{Tb}} template.
EDTA
Hi Ben. I hope you're doing well. I just wanted to let you know that there is a question at Talk:EDTA about one of the images you have created, File:EDTA-xtal-3D-balls.png. -- Ed (Edgar181) 13:09, 21 May 2010 (UTC)
- Hi Ed, long time no see! Thanks for the notification, I'll reply now.
- Cheers,
IUPAC Naming
I am confused over why the IUPAC names that I've been adding are being reverted, they are genuine and recommended by the IUPAC Provisional Recommendations for the Nomenclature of Organic Chemistry published in 2004. They are not generated by myself; and they in the majority sourced from the internet. Even though some are obscure, they are prefered IUPAC names as opposed to the traditional names. I'm at a loss for how I can possibly discuss the placement of IUPAC names in the article, as I'm breaking no wikipedia rules, but trying to add factual material. Almost in every case, my naming edits are reverted without explanation. I need to know why conformity to uniformity is being so strongly opposed in this case. If IUPAC nomenclature is not being acknowledged than why was it created. As a note, I'm also engaged with chemistry at a university.--Plasmic Physics (talk) 12:52, 4 June 2010 (UTC)
- This discussion is of interest to most Wikipedia chemistry editors, so I have moved it to Wikipedia talk:WikiProject Chemicals#user:Plasmic Physics.
- Hope this is OK with you,
When should I be expecting a preliminary draft of the guidelines?--Plasmic Physics (talk) 11:11, 9 June 2010 (UTC)
- I don't know, it depends on whether anyone writes them.
- For now, stick to IUPAC documents as references for IUPAC nomenclature.
There are none that I can find.--Plasmic Physics (talk) 00:32, 10 June 2010 (UTC)
- Try searching http://www.iupac.org/, http://www.acdlabs.com/iupac/nomenclature/, and http://www.chem.qmul.ac.uk/iupac/.
I already did, and I can tell you that they do not contain IUPAC names, only recommendations for how to construct the names.--Plasmic Physics (talk) 02:52, 10 June 2010 (UTC)
- You're probably best off waiting until preferred IUPAC names come into widespread usage.
So I can add it under Other Names without a reference then? PINs will take years to be in widespread use. I'm not actually talking about PINs, I'm talking about systematic names that are already in use to some degree, be they PINs or not.--Plasmic Physics (talk) 05:42, 10 June 2010 (UTC)
- Don't add anything without a reference. If an IUPAC name is in widespread use, you could cite an article from a mainstream peer-reviewed journal, or better yet a review article, or even more preferable, a good textbook.
- Ask again at WP:CHEM.
What about all other Other Names that are unrefferenced?--Plasmic Physics (talk) 12:42, 10 June 2010 (UTC)
- It's not ideal, but it's not a big problem. It's probably more effort to remove them, and many of them could easily be referenced, it just takes time and effort and isn't really worth it at the moment.
- Your best bet is to contribute other content from reliable sources. Ask WP:CHEM for advice, or try User:Smokefoot.

|
The Original Barnstar | |
| Awarded for making File:Sodium-selenide-unit-cell-1992-CM-3D-balls.png. It's beautifully made in depicting the unit cell of sodium selenide. The rhombic dodecahedral tiling is just tantalizing. Thanks for making my day today.—Tetracube (talk) 17:45, 11 June 2010 (UTC) |
WikiProject Bedfordshire
Hey,
- You are listed as a member of WikiProject Bedfordshire, a project that I am currently trying to revive. Please have a look at the projects talk page for an explanation. If you would still like to participate, please could you leave a note on my talk page. Thanks, Acather96 (talk) 20:10, 11 June 2010 (UTC)
WikiProject Bedfordshire

Hello, Benjah-bmm27, this message is being sent to inform you that, due to over three monts editing inactivity on WikiProject Bedfordshire, your username has been moved to an 'inactive participant'. If you object or would like to re-start contributing, please move your name back up to the active members list. Thanks,
Acather96 (talk) 07:41, 3 July 2010 (UTC)
Pharmacology stub templates
Hi! I don't know whether this interests you, but the new drug stub templates use your space filling models of various drugs, and they look absolutely fabulous. Cheers, ἀνυπόδητος (talk) 17:02, 3 July 2010 (UTC)
- They look great! Thanks very much for the message.
Probably not this time
Probably nothing will come of my suggestion to re-balance the distorted world-view of PProctor, and it would be ill-advised to go into the fray without the participation of a lot of other editors. I am not losing sleep over it. See you around.--Smokefoot (talk) 02:47, 6 July 2010 (UTC)
- OK, fair enough. Still ridiculous, though!
- Cheers,
chromium(VI) peroxide
There is some serious angle strain in this molecule. Also if it wouldn't probably spontaneously explode upon distillation, I wonder if it is a little volatile, like Titanium tetrachloride. Anyway, this is on top of a peroxy motif attached to Cr(VI). O-O bonds are usually long, but from that ball and stick image it seems to me like complexed dioxygen? John Riemann Soong (talk) 01:14, 6 August 2010 (UTC)
- I think it's more like O22− acting as a pi-donor ligand, much as ethylene does in, say, Zeise's salt. If you think about it like this, the peroxide ions are donating some of the electron density in their O-O π* antibonding molecular orbitals to chromium, so that should strengthen and shorten the O-O bond(s). If you fully removed two electrons from the two O-O π* MOs in O22−, you would indeed have O2. If you removed one electron from O22−, you'd get superoxide, O2−.
Heya
If you have time, could you work your magic and make a picture of this compound's crystal structure? The ref is in the article already, but I can't read more than the first page for now. Thanks! --Rifleman 82 (talk) 01:49, 9 August 2010 (UTC)
- Thank you! --Rifleman 82 (talk) 04:55, 9 August 2010 (UTC)
Hmm... it appears from the crystal structure that this is actually the isothiocyanate, not the thiocyanate? What does the article say? Thanks! --Rifleman 82 (talk) 04:57, 9 August 2010 (UTC)
- The article just describes what you can see in the picture, really. Says the N-Co and S-Co interactions are predominantly ionic.
- What do you make of that?
Nitrate-ion
Do you mind if I replace the δ symbols with the real charges in 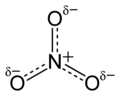 , or do you want to dit yourself? Use of that symbol for specific charges is not accepted by IUPAC and might be confusing for many readers.--Wickey-nl (talk) 14:36, 30 August 2010 (UTC)
, or do you want to dit yourself? Use of that symbol for specific charges is not accepted by IUPAC and might be confusing for many readers.--Wickey-nl (talk) 14:36, 30 August 2010 (UTC)
You can't replace it with three negative charges... it won't make sense. --Rifleman 82 (talk) 14:58, 30 August 2010 (UTC)
- I could replace the δ symbols with one-third negative charges if you like.
- Ben (talk) 15:05, 30 August 2010 (UTC)
- Thanks, with two-third negative. Nitrogen is 1+. Nitrate.--Wickey-nl (talk) 15:12, 30 August 2010 (UTC)
Here it is: File:Nitrate-ion-resonance-hybrid-2D.png.
Ben (talk) 19:49, 31 August 2010 (UTC)
- I am afraid you did not get the clue. Mixing of formal and partial charges in one structure is not logical, because the letter are known; and conflicting with the IUPAC rules, p.378. It had been better to adapt the original file. You can also make N δ+, but that is not useful.--Wickey-nl (talk) 13:18, 5 September 2010 (UTC)
What're you talking about?. We don't always follow IUPAC rules. We make diagrams that illustrate the points we want to make. Comparing the original and new images, you can see the relation between them: δ = ⅔.
Ben (talk) 16:22, 5 September 2010 (UTC)
- You are free to make useless images.--Wickey-nl (talk) 15:25, 7 September 2010 (UTC)
What kind of diagram do you want?
You asked me to make one, so I did. Then you say I didn't get the clue - what clue? If you want people to do things for you, be civil.
Ben (talk) 16:39, 7 September 2010 (UTC)
- I understand it was sounding unkind. The new image was just fine and much more useful than the old one. But it is a good custom to follow IUPAC rules, unless you have a good reason to do not. If there is no good reason, the image is more likely harmful.--Wickey-nl (talk) 15:31, 8 September 2010 (UTC)
OK, fair enough. I tend to keep images rather than replace them, unless they are factually incorrect. I think many people have too much respect for IUPAC. Their rules aren't always good ones.
There are different opinions on the matter, of course, but it does little harm to have several images showing the same idea in different ways.
If you feel strongly about δ symbols and full charges in the same image, raise the issue at WP:CHEMISTRY.
Cheers,
Ben (talk) 15:39, 8 September 2010 (UTC)
NaSH
Nice work on the hydrosulfide structure. Probably can be used for KSH. And thank you for helping repair the connection to redistribution (chemistry). Best wishes,--Smokefoot (talk) 17:08, 18 September 2010 (UTC)
- No problem. I'm trying to get three images, one for each modification, but disorder is hard to depict without confusing non-specialists.
- Good work cleaning up molecular diagrams of non-molecular substances!
- Ben (talk) 17:24, 18 September 2010 (UTC)
- Well, I think between the two of us, Wikipedia has unpropagated a lot of misconceptions about what stuff looks like. But it is also possible that readers just ignore the complicated images and the text and are still convinced that silver chloride is a pale green gas, etc. But the work goes on against these infidels...--Smokefoot (talk) 20:55, 18 September 2010 (UTC)
Accelrys
I have the picture (3D) model of a chemical compound made with Accelrys, but I don't have Photoshop. What can I do? YOSF0113 (talk - contributions) 05:26, 19 September 2010 (UTC)
- Any image editor will work fine, there are free ones like GIMP.
- If you need any more help, just ask.
- Cheers,
Au2S
Nice image for Au2S? I found about 100 papers through a CAS search, but no recent X-ray report. If you ever run across anything real on Au2S3, we might consider starting that article or at least mentioning it in gold(I) sulfide. I am trying to find material on isolation of [Au(SO3)2]3-, which is S-bonded, and is used as an alternative to [Au(CN)2]- for electroplating Au.--Smokefoot (talk) 23:50, 3 October 2010 (UTC)
- I've had a look for crystal structures containing [Au(SO3)2]3-, but to no avail so far. As for Au2S3, this 2003 paper suggests it's amorphic.
I added the MO story to the enolate section. I hope you approve... Of course all electron pushers will hate me, but so it goes. ;-) (Sorry I am inorganic/soldi state). Hmm, Au2S? I don;t think it exists as stable compound and yes in a previous life I did some work on the Ag2S-Au2S system.. But that is long ago. Greetings Jcwf (talk) 19:44, 6 October 2010 (UTC)
- Looks great, thanks very much.
- I can quite believe that Au2S3 doesn't exist, but Au2S certainly seems to. See Gold(I) sulfide for references to Greenwood & Earnshaw, etc. On the image description page of the unit cell I recently added to that article, I've provided a link to the original paper: Solid State Ionics (1995) 79, 60-65.
- If you think there's something wrong with this, please reply! I'll make any necessary corrections.
- Cheers,
One way to measure sulfur gas pressures is to exposure a series of Ag-Au alloys (electrum) to the vapor and look at which one gets tarnished. The more silver the sooner it tarnishes. You can certainly go to AuAgS with that (that is a stable compound) but more gold rich than that at some point it stops getting tarnished even at high sulfur vapor pressures. If memory serves I think there is thermodynamic evidence from that at the end member (Au itself) should not give a stable sulfide anymore. Of course it may well be that you can make Au2S as a metastable phase somehow from other sources than the elements. Judging from the higher temperature phases in the Ag2-Au2S system that are all cubic, you'd expect a bcc sulfur lattice with Au on four of the 1/8 1/8 1/8 positions and at cursory glance that is what you are showing I think.
Jcwf (talk) 21:55, 6 October 2010 (UTC)
- Oh wow - quite a cheeky little system then.
- I'll have a read about it, see if there are any new developments.
- Thanks for the info :)
- I wrote up some stuff about it for our cumes at some point See. Not about the stability but our paper is referenced there.
Jcwf (talk) 17:20, 7 October 2010 (UTC)
image request
3M's PFOA replacement is ADONA, ammonium 4,8-dioxa-3H-perfluorononanoate, CF3OCF2CF2CF2OCHFCF2COO−NH4+. Would you make an image? Thanks! -Shootbamboo (talk) 22:50, 21 October 2010 (UTC)
- 300px - how's that?
- Cheers,
- Ben (talk) 12:15, 22 October 2010 (UTC)
- It's great, thanks. =) Here it is in the page. Thanks again. -Shootbamboo (talk) 18:19, 31 October 2010 (UTC)
Very good news
Dont know wether you were aware or not, but [3] has been included in The Big Book of Top Gear 2011. Acather96 (talk) 07:03, 31 October 2010 (UTC)
- Awesome, thanks for letting me know!
You're fallible
Hi over there. Could you edit this file if you still have it?
Cheers, --Smokefoot (talk) 20:57, 14 November 2010 (UTC)
- Done.
- Thanks for reminding me about that one!
image request
These are not like my previous requests, but graphane and fluorographene, perhaps? Thanks again for your good work here. =) Shootbamboo (talk) 22:10, 16 November 2010 (UTC)
- A picture was posted at fluorographene so nevermind. -Shootbamboo (talk) 19:43, 27 November 2010 (UTC)
Sorry mate, I wanted to make an image but it was a bit of a technical challenge.
Ben (talk) 20:29, 27 November 2010 (UTC)
- No problem at all. Thanks again. -Shootbamboo (talk) 05:16, 28 November 2010 (UTC)
Request for assistance - image from crystal structure data
Hi Ben, I know you do some work with chemical structures, which I am hoping includes crystal structure data. I recently got the article Hans Freeman to GA standard, and am considering a run for FA. Freeman's PhD work included the crystal structure of biuret hydrate (completing most calculations by hand!), and I think an illustration based on the Acta Cryst structure (ref'd in the article) would be a nice addition. Leyo suggested you might be able to help. Would you be able to provide a suitable image? Alternatively, can you suggest someone else who would be better suited to help? Thanks. EdChem (talk) 12:46, 22 November 2010 (UTC)
- Hi Ed, I've located the structural data required. Give me a day or two and I'll have something for you.
- Cheers,
Hexacyclinol
I saw the chemical structure of hexacyclinol that you drew on your website. Would it be possible for you to draw the structure Grafe proposed (see http://pubs.acs.org/cen/news/84/i31/8431notw1.html) which was ultimately shown to be incorrect and put both of them on Commons? I'm working on putting an article together for this compound. Thanks, shoy (reactions) 18:10, 7 December 2010 (UTC)
- User:EdChem pointed out at Template_talk:Did_you_know#Articles_created.2Fexpanded_on_December_22 that your structure for hexacyclinol was incorrect. Just a heads-up. shoy (reactions) 17:48, 23 December 2010 (UTC)
Thanks for the heads up, I've fixed the image on my website and here.
Cheers,
Ben (talk) 19:31, 23 December 2010 (UTC)
- Looks like File:(+)-hexacyclinol-2D-skeletal-200px.png (the one missing the side-chain) is unused and doesn't seem useful (due to that structural flaw). Maybe tag it for deletion? DMacks (talk) 22:54, 2 January 2011 (UTC)
Absolutely, fire away.
Ben (talk) 17:17, 3 January 2011 (UTC)
Looks like it's gone. Thanks for the quick work, guys. shoy (reactions) 18:02, 5 January 2011 (UTC)
Hexol
Hi Ben, would a new image of hexol for Coordination complex and Hexol be a good idea? If you like and have time.--Wickey-nl (talk) 08:39, 8 December 2010 (UTC)
- Done. Sorry for the month-long wait!
- Cheers,
Opening a .mol file in Accelrys Discovery Studio Visualizer
Hi, I have problems with opening a .mol file in Accelrys Discovery Studio Visualizer 2.5.5. When I open the file through DSV, I can see the data but I cannot see the 3D model in the Visualizer. Can you help me? YOSF0113 (talk - contributions) 05:50, 12 December 2010 (UTC)
Talk by Jimbo
Hi Ben, have you seen that Jimmy Wales is giving a talk in Bristol next week? If you want to come, I think there are still some tickets left for wikimedians, I think you just need to email the address in the link, or if that doesn't work, drop Steve Virgin an email. I think there may be a meet up too, check out here for more details. Happy New Year SmartSE (talk) 17:08, 5 January 2011 (UTC)

Hey Ben. I have one question regarding your picture about the synthesis fo 1,4-dioxin. From structure 2 to 3 one additional oxygene is appearing (epoxide). Would it be helpful to indicate this process by adding somethink like [O]? Cheers, --Yikrazuul (talk) 19:00, 12 January 2011 (UTC)
- Good idea, I'll do it ASAP.
- Thanks for the suggestion,
- Niceeeee. --Yikrazuul (talk) 20:12, 13 January 2011 (UTC)
Hey Ben, I just e-mailed you at the e-mail address provided by your website. (ben @ ben-mills...blahblahblah) — Preceding unsigned comment added by SubDural12 (talk • contribs) 06:44, 2 February 2011 (UTC)
A15 phases
A15 phases needs an image similar to the Wurtzite-unit-cell-3D-balls.png. The structure is available on this page. It is the structure for β-Tungsten and for Nb3Sn a important type II superconductor. Is it possible for you to make a image for me? Thanks stone.--Stone (talk) 11:48, 5 February 2011 (UTC)
- No problem, I'll do it now.
- Thanks! One of the image shows very nicely the chains of Nb which are credited with the superconductivity! Thanks!
--Stone (talk) 17:45, 5 February 2011 (UTC)
presently versus currently
I have two printed copies of the OED, and the computer version. I consult it often, and depend on it to help me find the right word, particularly when writing historical fiction. The "problem" with the OED is that it's a descriptive dictionary. Unlike, say, the official French dictionary, it is neither prescriptive nor proscriptive. It simply records English as it is written.
This means it includes bad usage. "Presently" has traditionally meant "soon", not "now". I'm a nut about sticking with traditional usage, because the blurring of meanings makes English less expressive. I don't object to adding words, as long as there isn't an existing word that conveys the same sense. WilliamSommerwerck (talk) 15:03, 5 February 2011 (UTC)
- Bad is a subjective term. Many words have had different meanings in the past. Why should your prescriptive view of language be considered superior to a more descriptive view? Especially with such a well-established usage of "presently". You may feel that the evolution of the meanings of various words in English renders the language less expressive, but many others do not.
- To avoid antagonising other editors, I recommend replacing edit summaries such as
with something less dogmatic like"Presently" and "currently" DO NOT mean the same thing.
"Presently" can mean "soon" as well as "now". I'm replacing it with "currently", which is unambiguous.
EDTA crystal structure
commons:File:EDTA-xtal-3D-sticks.png is a great example of zwitterions! The analogous commons:File:EDTA-xtal-3D-balls.png appears to be missing a hydrogen on the upper-right carboxyl. I can't seem to access the xray coordinates...did they actually find the H atoms and know the specific protonation sites, or just add them where they are expected based on geometry and symmetry? DMacks (talk) 18:02, 5 February 2011 (UTC)
- Hi DMacks,
- I think the hydrogen atom you speak of is there, just obscured. I can re-draw it.
- I just checked the database, and the hydrogen atoms were located. The carboxylic acid OH groups adopt that unusual conformation due to hydrogen bonding with the -CO2− group of a neighbouring molecule. The hydrogen atom sits almost halfway between the two groups:
- RC(=O)O···H···O(O=)CR
- I could redraw the molecule, switching round which carboxyl groups are ionised and which are not, that would lead to a clearer structure.
- What do you think?
- Ben (talk) 18:55, 5 February 2011 (UTC)
- Maybe could just tilt the -balls structure backward a bit so that H is not hidden behind its O? I have no thoughts on use of the "other" proton-attachments, just trying to make the structures more visibly agree, and preserve the nice C2 symmetry. DMacks (talk) 20:22, 5 February 2011 (UTC)
Poor quality images
What do YOU do when you see very poor quality images of molecules?
I'd like to remake some 3-d structures that I have seen before, but I do not know if it is unethical? Hell, I don't really think it would be wrong, if the images really are poor quality.
This image isn't incorrect in structure, just low in quality.
After all, the integrity of Wikipedia must be preserved over the integrity of any one user, no?
SubDural12 (talk) 04:23, 11 February 2011 (UTC)
- I remake them!
- It's not unethical at all.
- Do you have any particular images in mind?
I changed the image of hydrocodone. Turns out he had the structure wrong as well. Pubchem documented two of the rings being structurally different than that of which he had.
-
Previous
-
Current
SubDural12 (talk) 19:44, 11 February 2011 (UTC)
- File:Hydrocodone 3d balls.png isn't low quality. Plus I think you have the wrong enantiomer. Ball-and-stick models show the difference much more clearly.
- PubChem isn't the best place to get structural information, and you have to correctly interpret what you do find.
- If you make a ball-and-stick or stick image of your model, I can check it for you.
Damn, I felt really good about PubChem. I thought I was finally getting some access to a really good site. How can I confirm these isomers myself? And why on earth is PubChem posting these biologically inactive forms?
SubDural12 (talk) 00:40, 12 February 2011 (UTC)
Hey, I fixed hydrocodone2.png & hydrocodone3.png, and I also fixed oxycodone2.png & and oxycodone3.png.
I do not wish to replace your image of hydrocodone, but I was hoping that you would check mine anyways, just for the sake of validity. I was hoping to use my model of oxycodone on the main page oxycodone though, so please let me know if I have screwed something up again.
P.s. I found out that my searching of "hydrocodone" on pubmed was the reason for my ill results. I should have instead, searched for "dihydroxycodeinone." This returns some more alternate isomers.
SubDural12 (talk) 01:29, 12 February 2011 (UTC)
- Hi SubDural, File:oxycodone3.png is correct, I compared it with the crystal structures of some oxycodone derivatives. I searched PubChem for "hydrocodone" and all the hits had the correct stereochemistry - do you know how to interpret wedged and hashed bonds in skeletal formulae?
- One thing I've noticed in your images, for example hexazine, is your aromatic rings have some bonds shorter than others. They should all be the same length: see benzene for an explanation.
@SubDural12: If you are looking for images of molecules that need to be redrawn, you will find some in Commons:Category:Low quality chemical diagrams/expired. --Leyo 22:20, 12 February 2011 (UTC)
- @ Leyo: Thank you very much, I appreciate it.
- @ Ben: I haven't noticed the odd uneven bond lengths in my aromatic rings. They seem to be correct and even in BKchem, and then they translate into DSV oddly. I will have to experiment to see what the problem is.
SubDural12 (talk) 04:22, 13 February 2011 (UTC)
- I found out what the problem is: I was creating alternating double bonds in BKchem, transferring into DSV, and THEN changing the bonds into aromatic bonds. However, DSV doesn't automatically update bond lengths. I found out that the best way is to draw the molecule with aromatic bonds prior to transferring into DSV. Then the bond lengths are all equal.
- Wow, HOW DID YOU NOTICE THAT?!?!?! I am amazed, they all seemed equal length to me. Nice job, man! THANKS!!
SubDural12 (talk) 04:40, 13 February 2011 (UTC)
Best way to contact you
Ben,
What is the best way to contact you? I would hate to flood your talk page with my incessant rambling. However, until then, I have a question for you involving my structure of quisqualic acid. I have found a local university library to go to in my spare time, and they have a lot of access to research journals. I now have access to Acta Crystallographica, The Journal of Organic Chemistry, and many more great journals. It was a bit confusing to interpret this data, though, and I hope that I interpreted it correctly. I have posted the updated image of quisqualic acid on the wikipedia page. It is a zwitterion, like you previously mentioned.
SubDural12 (talk) 23:51, 14 February 2011 (UTC)
- Hi SubDural, it's fine to contact me on this page. That's what it's for!
- The image looks fine to me. Was it this paper (Acta Cryst. B (1976) 32, 951-953) you got the structure from?
You got it! That's the one.
I was curious if you had ever found N-methyl-D-aspartic acid before? I found N-methyl-D-aspartic acid monohydrate, but the hydrogen bonds alter the structure between the two molecules.
I also could not find L-ornithine.
SubDural12 (talk) 19:55, 15 February 2011 (UTC)
copper acetate
The image you created File:Copper(II)-acetate-3D-balls.png needs to be modified slightly. The image is used in a new article magnetochemistry which makes it clear why there is no Cu-Cu bond. If you are able to make the necessary changes, please go ahead. Many thanks. Petergans (talk) 15:45, 20 February 2011 (UTC)
- I have graphically quickfixed the structure here, with a reference. Ben, if you redraw it with a proper program, just overwrite my image file. Materialscientist (talk) 00:39, 22 February 2011 (UTC)
Hi Ben, some years ago you have drawn a structural formulae for the box. However, the absolute configuration is missing in two positions. Best regards, --Jü (talk) 16:57, 21 February 2011 (UTC)
Hey Ben
I looked through the wiki markup and it seems fine but for some reason your image is not displaying properly... Perhaps you can take a look. --Rifleman 82 (talk) 01:38, 3 April 2011 (UTC)
- There are some serious wikipedia database update lags there days, right now for example. Materialscientist (talk) 01:57, 3 April 2011 (UTC)
Well dont abandon us!
Other editors must share your sense of exasperation over the persistent irresponsible/nutty behavior. You have to admit that most pages are much better than when you and I started. Fewer real embarrassments. Readers now must understand that most inorganic solids have polymeric structures. So for the annoying stupidity, including university-driven content, we just grind away at the worse parts when these editors cease at the end of their term. So dont lose hope. And be resigned to the fact that we're always going to have this kind of strangeness.--Smokefoot (talk) 18:02, 16 April 2011 (UTC)
odd looking chembox
At DOI the infobox looks odd under the Jmol images section. A quick look from me didn't spot anything. Might be a quick fix can you take a look? Thanks. Shootbamboo (talk) 00:12, 17 April 2011 (UTC)