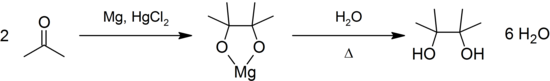Pinacol is a branched alcohol which finds use in organic syntheses. It is a diol that has hydroxyl groups on vicinal carbon atoms. A white solid that melts just above room temperature, pinacol is notable for undergoing the pinacol rearrangement in the presence of acid and for being the namesake of the pinacol coupling reaction.
Preparation
It may be produced by the pinacol coupling reaction from acetone:[1]
Reactions
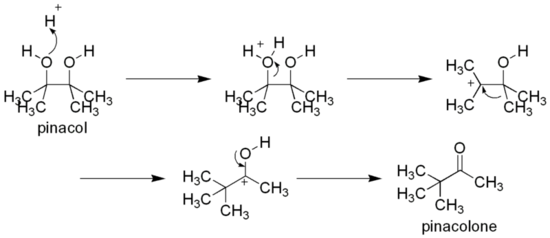
As a vicinal diol, it can rearrange to pinacolone by the pinacol rearrangement, e.g., by heating with sulfuric acid:[2]
Pinacol can be used with borane and boron trichloride to produce useful synthetic intermediates such as pinacolborane, bis(pinacolato)diboron,[3] and pinacolchloroborane.
See also
References
- ^ Roger Adams and E. W. Adams. "Pinacol Hydrate". Organic Syntheses; Collected Volumes, vol. 1, p. 459.
- ^ G. A. Hill and E. W. Flosdorf (1941). "Pinacolone". Organic Syntheses; Collected Volumes, vol. 1, p. 462.
- ^ Tatsuo Ishiyama; Miki Murata; Taka-aki Ahiko; Norio Miyaura (2004). "Bis(pinacolato)diboron". Organic Syntheses; Collected Volumes, vol. 10, p. 115.