The Knorr quinoline synthesis is an intramolecular organic reaction converting a β-ketoanilide to a 2-hydroxyquinoline using sulfuric acid. This reaction was first described by Ludwig Knorr (1859–1921) in 1886[1]

The reaction is a type of electrophilic aromatic substitution accompanied by elimination of water. A 1964 study found that with certain reaction conditions formation of a 4-hydroxyquinoline is a competing reaction.[2] For instance, the compound benzoylacetanilide (1) forms the 2-hydroxyquinoline (2) in a large excess of polyphosphoric acid (PPA) but 4-hydroxyquinoline 3 when the amount of PPA is small. A reaction mechanism identified a N,O-dicationic intermediate A with excess acid capable of ring-closing and a monocationic intermediate B which fragments to aniline and (ultimately to) acetophenone. Aniline reacts with another equivalent of benzoylacetanilide before forming the 4-hydroxyquinoline.

A 2007 study[3] revised the reaction mechanism showing that based on NMR spectroscopy and theoretical calculations an O,O-dicationic intermediate (a superelectrophile) is favored comparing to the N,O dicationic intermediate. For preparative purposes triflic acid is recommended:
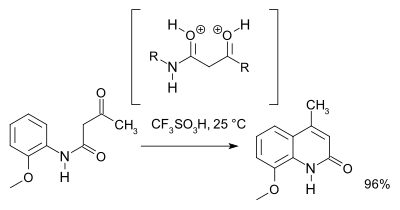
References
- ^ Synthetische Versuche mit dem Acetessigester Justus Liebig's Annalen der Chemie Volume 236, Issue 1–2, Date: 1886, Pages: 69–115 Ludwig Knorr doi:10.1002/jlac.18862360105
- ^ The Conversion of Benzoylacetanilides into 2- and 4-Hydroxyquinolines B. Staskun J. Org. Chem. 1964; 29(5); 1153–1157. doi:10.1021/jo01028a038
- ^ Knorr Cyclizations and Distonic Superelectrophiles Kiran Kumar Solingapuram Sai, Thomas M. Gilbert, and Douglas A. Klumpp J. Org. Chem. 2007, 72, 9761–9764 doi:10.1021/jo7013092








