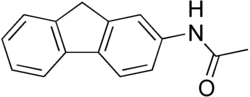
Hydroxyacetylaminofluorene
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
N-(9H-Fluoren-2-yl)-N-hydroxyacetamide | |
| Other names
N-Hydroxy-2-acetamidofluorene; Hydroxyfluorenylacetamide
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| KEGG | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C15H13NO2 | |
| Molar mass | 239.27 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Hydroxyacetylaminofluorene is a derivative of 2-acetylaminofluorene used as a biochemical tool in the study of carcinogenesis.
Biochemistry
N-hydroxy-2-acetamidofluorene reductase is an enzyme that catalyzes the conversion of the carcinogen, hydroxyacetylaminofluorene to 2-acetylaminofluorene. It uses reduced nicotinamide adenine dinucleotide (NADH) as its cofactor.[1][2]
See also
References
- ^ Gutmann HR, Erickson RR (1969). "The conversion of the carcinogen N-hydroxy-2-fluorenylacetamide to o-amidophenols by rat liver in vitro. An inducible enzymatic reaction". J. Biol. Chem. 244 (7): 1729–40. doi:10.1016/S0021-9258(18)91744-8. PMID 5780838.
- ^ Kitamura S, Tatsumi K (1985). "Purification of N-hydroxy-2-acetylaminofluorene reductase from rabbit liver cytosol". Biochem. Biophys. Res. Commun. 133 (1): 67–74. Bibcode:1985BBRC..133...67K. doi:10.1016/0006-291X(85)91842-X. PMID 4074379.