Hexafluoroacetone (HFA) is a chemical compound with the formula (CF3)2CO. It is structurally similar to acetone; however, its reactivity is markedly different. It is a colourless, hygroscopic, nonflammable, highly reactive gas characterized by a musty odour.[2] According to electron diffraction, HFA and acetone adopt very similar structures, the C-O distance being only longer in the fluorinated compound (124.6 vs 121.0 pm), possibly due to steric effects.[3]
The term "hexafluoroacetone" can refer to the sesquihydrate (1.5 H2O), which is a hemihydrate of hexafluoropropane-2,2-diol (F
3C)
2C(OH)
2, a geminal diol. Hydrated HFA behaves differently from the anhydrous material.
Synthesis
The industrial route to HFA involves treatment of hexachloroacetone with HF (a Finkelstein reaction):[4]
- (CCl3)2CO + 6 HF → (CF3)2CO + 6 HCl
Laboratory methods
Hydrated HFA can be converted to HFA by treatment with hot sulfuric acid.[5]
It has also be prepared from hexafluoropropylene oxide, which will rearrange to give HFA when heated in the presence of a Lewis acid such as AlCl3.[6] The Lewis acid catalysed oxidation of hexafluoropropylene will also produce HFA, via a similar mechanism.
Although it is commercially available, HFA can be prepared on the laboratory-scale from hexafluoropropylene.[7] In the first step KF catalyzes the reaction of the alkene with elemental sulfur to give the 1,3-dithietane dimer of hexafluorothioacetone. This species is then oxidized by potassium iodate to give HFA.[8]
Uses
Hexafluoroacetone is used in the production of hexafluoroisopropanol:
- (CF3)2CO + H2 → (CF3)2CHOH
It is also used as a precursor to hexafluoroisobutylene,[4] a monomer used in polymer chemistry, and as a building block in the synthesis of midaflur, bisphenol AF, 4,4′-(hexafluoroisopropylidene)diphthalic anhydride, and alitame.
Reactivity
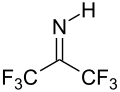
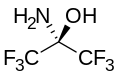
With water, hexafluoroacetone converts to the hydrate. The equilibrium constant (Keq) for the formation of this geminal diol is 106 M−1. The analogous equilibrium for acetone is an unfavorable 10−3 M−1.[9] Hexafluoroacetone-hydrates are acidic. In an analogous reaction, ammonia adds to hexafluoroacetone to give the hemiaminal (CF3)2C(OH)(NH2) which can be dehydrated with phosphoryl chloride to give the imine (CF3)2CNH.[10]
Nucleophiles attack occurs at the carbonyl carbon of Hexafluoroacetone, as illustrated above. Thus, HFA readily forms lactones when treated with hydroxy- and amine-substituted carboxylic acids. In such reactions, HFA serves both as electrophile and dehydrating agent:[5]
- RCH(OH)CO2H + O=C(CF3)2 → RCH(O)CO2C(CF3)2 + (HO)2C(CF3)2
See also
References
- ^ a b c d e f NIOSH Pocket Guide to Chemical Hazards. "#0319". National Institute for Occupational Safety and Health (NIOSH).
- ^ CDC - NIOSH Pocket Guide to Chemical Hazards
- ^ Hilderbrandt, R. L.; Andreassen, A. L.; Bauer, Simon Harvey (1970). "Electron diffraction investigation of hexafluoroacetone, hexafluoropropylimine, and hexafluoroisobutene". The Journal of Physical Chemistry. 74 (7): 1586–1592. doi:10.1021/j100702a030.
- ^ a b Günter Siegemund; Werner Schwertfeger; Andrew Feiring; Bruce Smart; Fred Behr; Herward Vogel; Blaine McKusick (2002). "Fluorine Compounds, Organic". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a11_349.
- ^ a b Spengler, Jan; Böttcher, Christoph; Albericio, Fernando; Burger, Klaus (2006). "Hexafluoroacetone as Protecting and Activating Reagent: New Routes to Amino, Hydroxy, and Mercapto Acids and Their Application for Peptide and Glyco- and Depsipeptide Modification". Chemical Reviews. 106 (11): 4728–4746. doi:10.1021/cr0509962. PMID 17091933.
- ^ Millauer, Hans; Schwertfeger, Werner; Siegemund, Günter (March 1985). "Hexafluoropropene Oxide — A Key Compound in Organofluorine Chemistry". Angewandte Chemie International Edition in English. 24 (3): 161–179. doi:10.1002/anie.198501611.
- ^ Anello, Louis G.; Van der Puy, Michael (January 1982). "A convenient synthesis of hexafluoroacetone". The Journal of Organic Chemistry. 47 (2): 377–378. doi:10.1021/jo00341a046.
- ^ Van Der Puy, M.; Anello, L. G. (1985). "Hexafluoroacetone". Organic Syntheses. 53: 154. doi:10.15227/orgsyn.063.015.
- ^ Lemal, David M. (2004). "Perspective on Fluorocarbon Chemistry". The Journal of Organic Chemistry. 69 (1): 1–11. doi:10.1021/jo0302556. PMID 14703372.
- ^ W. J. Middleton; H. D. Carlson (1970). "Hexafluoroacetone imine". Org. Syntheses. 50: 81–3. doi:10.15227/orgsyn.050.0081..









You must be logged in to post a comment.